Potassium fluoborate is also critical in the production of boronizing steel – a very hard steel that offers superior wear resistance that is commonly used in oil-and-gas fields within drilling equipment (Fig. Potassium fluoborate, a white crystalline salt, is an important ingredient in grain-refining salts for aluminum and can be used either separately or combined with potassium-titanium-fluoride to form a grain-refining flux, which can improve the physical properties in aluminum castings as well as prevent cracking. Its most common use is as a fertilizer, called potash. Potassium-chloride salt is used for smelting, hardening and soldering as well as for surface treatment and galvanizing processes. Potassium is an important element in the field of metallurgy. Davy’s young assistant, famed English scientist Michael Faraday, went on to become one of the most influential figures in the history of science.
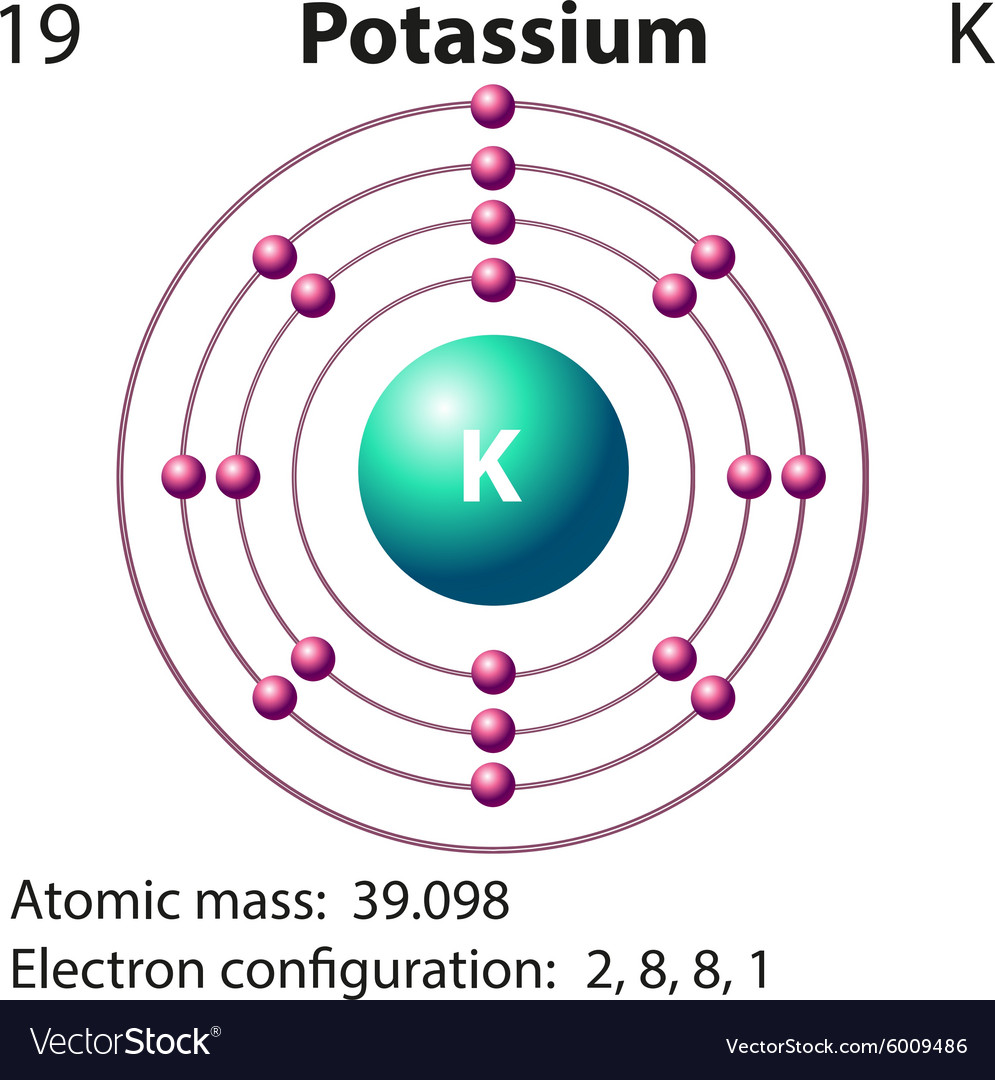
He performed electrolysis on very dry, molten caustic potash (KOH, potassium hydroxide) and noted that potassium collected at the cathode. The electrolysis technique allowed him to isolate several substances for the first time, including potassium. Davy had taken an interest in Italian scientist Alessandro Volta’s invention of the battery in 1800 and recognized its value in what became to be known as the field of electrochemistry, of which Davy is considered the founder. Potassium was first isolated in 1807 by Cornish chemist and inventor Sir Humphry Davy through the use of electrolysis. 1) is never found in nature and instead can be produced by electrolysis of potassium chloride or hydroxide. Due to its reactivity, pure potassium (Fig. In fact, potassium shares many of the same properties as sodium, such as being lighter than water and the ability to float. A similar method is used to store pure sodium. For this reason, pure potassium is stored submerged in mineral oil. It oxidizes rapidly in air and reacts aggressively with water to form hydrogen, which can then be ignited by the heat generated in the reaction. Potassium is a soft, silvery metal with a waxy consistency that can be easily cut with a knife.
